
|
Boron Predeposition Diffusion
The boron predeposition transfers boron from a solid source to the wafer.
Boron, in the form of B2O3, diffuses from the source to the
wafer. When it reaches the silicon, it will undergo the following chemical reaction:
2B2O3 + 3Si → 3SiO2 + 4B (borosilicate glass)
The borosilicate glass (BSG) is in contact with the silicon surface. A concentration
gradient is formed, and since this process occurs at high temperature, diffusion
will occur. The surface concentration will be fixed at the solid solubility, and
the distribution will be in the form of a complimentary error function:
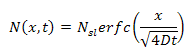
where:
N = concentration (cm-3)
Nsl = solid solubility limit for dopant (cm-3)
x = position inside silicon relative to the surface
D = diffusion coefficient for dopant (cm2/s)
t = time (seconds)
Equipment
Lindbergh-Tempress 8500 manual oxidation furnace chamber 8D
Supplies
- BN solid source: the BN wafer was oxidized at the beginning of the semester
- Solid source wafer boat
Operating parameters
- Furnace temperature: 950 °C
- gasses used: N2
- N2 flow
- standby: 100
- processing: 100
- Predeposition time: 15 min
Equipment/controls/tools locations
- Temperature controller: on the side of the furnace
- Gas panel: bottom rotameter at the front of the furnace
- Quartz handling: covered cart is to the left of the furnace, tongs are inside
- Boat: at the center of the furnace
Operating precautions
High temperatures
Use the high temperature gloves when handling hot equipment.
Contamination issues
- Quartzware is easily contaminated by alkali ions. This leads to premature quartz
failure (breakage) due to devitrification as well as unstable MOSFET Vt.
Once quartz is contaminated, little can be done to remove the contamination.
- Always wear latex gloves when working with the furnace.
- N2 should always be flowing in standby to minimize contamination by backstreaming
of air in the room into the hot chamber.
Operating procedure
- Degrease the wafer.
- Perform a 10-15 second etch in 50:1 DI:HF on your wafer if it has been more than
an hour since opening the diffusion windows, DI rinse, and N2 dry.
- Check the boron predep furnace and support equipment (i.e., gas flows and temperature).
The boron predep furnace should be at
950°C.
- Follow the procedure for furnace loading in appendix G of the paper version. Use
the Boron predep furnace and load the wafer so that the patterned side is facing
the nearest BN wafer. Be sure to record which position your wafer is in (see Appendix
G.4).
- After a
15
min predeposition at
950°C, unload your wafer.
- Use the LDS four-point probe to get a rough idea of the sheet resistance. Consult
the instructor if it's outside the range specified by the SPC chart, you may have to return
the wafer to the furnace.
Verify dopant type by using the hot point probe (Appendix E).
Rs=__________ Ω/square.

Note: The BN source transfers boron to the wafer via B2O3. The B2O3
reacts with the silicon to form a heavily doped SiO2 layer (borosilicate
glass), with a B:Si alloy layer at the BSG:Si interface. The BSG is easily removed
with 50:1 DI:HF, but the B:Si layer must be oxidized chemically before it can be removed
with the 50:1 DI:HF.
This transfer of boron using B2O3 is the ideal case for the
ECE444 lab, but can be greatly accelerated by the presence of H2 or H2O.
The hydrogen reacts with B2O3 to form HBO2 (meta-boric
acid), which has a vapor pressure much higher than B2O3. The
higher vapor pressure of the metaboric acid accelerates the growth rate of BSG,
therefore requiring a longer BSG etch.
|
|
IC Process
1.
2.
3.
4.
5.
6.
7.
8.
9.
10.
11.
12.
13.
14.
15.
16.
17.
18.
19.
20.
21.
22.
23.
24.
Answers provided by this service may not be relevant to the materials presented in this website.
|